**UPDATE 2021**
The TGA have reviewed all implants for use in Australia and have suspended the use of Grade 3 and 4 Macrotextued implants and some Microtextured implants associated with higher instances go BIA-ACLC.
It is important to note that the TGA has advised that removing implants as a precautionary measure is not required as 99.9% of patients with Microtextured implants will not develop BIA-ACLC. If you have symptoms of BIA-ACLC it is important to see your Doctor as soon as possible.
The Microtextured implants offered at Absolute Cosmetic are not within the list of suspended implants.
More information about the most recent decision from the TGA is available here. Information from the Australasian College of Cosmetic Surgery is available here.
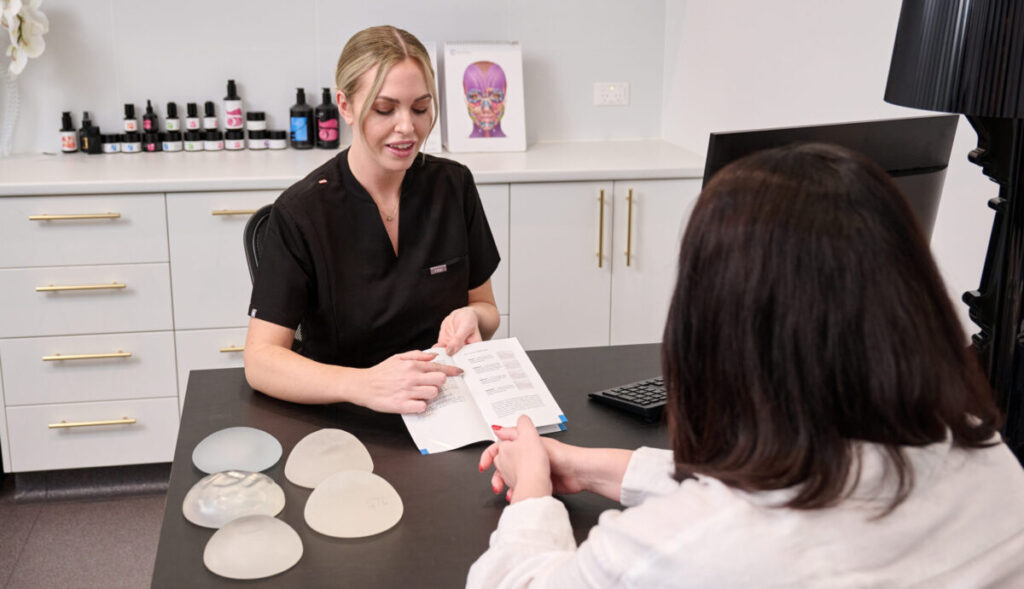
To arrange a complimentary Breast Implant Check, visit our page here, for more details, or call 9389 9099 to book a consult.
The TGA has announced that they are reviewing the use of a number of textured implants. Their proposed actions are to cancel or suspend the use of a number of implants due to the link between textured implants and BIA-ALCL.
BIA-ALCL is a rare cancer of the immune system that develops in the fluid and scar tissue around the implants. The symptoms usually involve a swelling of the breast, typically 3 to 14 years after the implantation. Less commonly, BIA-ALCL can take the form of a lump in the breast or the armpit.
It is a rare condition that is estimated to affect between 1 in 1,000 and 1 in 10,000 people with implants (figures from TGA). The vast majority of cases have been cured by the removal of the implant and the capsule surrounding the implant. There have been reports of fatalities from BIA-ALCL, however the number of fatalities is too small to be reliably calculated, it is considered to be less than 1 in 1 million.
The proposed changes affect textured implants only. At the time of publishing this post, no decision has been made, changes are proposed only, a decision will be made on the 24th July. The proposed cancellation and recall includes a range of Allergan Natrelle textured implants. The proposed suspension includes a range of implants from multiple suppliers including; Nagor, Emagin, Emergo, Cristaline, Polytech (polyurethane) and Mentor. The full list of proposed cancelled, suspended and imposition of conditions are available at https://www.tga.gov.au/alert/breast-implants-and-anaplastic-large-cell-lymphoma.
Please note that if proposed changes of recall come into effect, it is recalling implants that have not yet been implanted. The TGA states that “There is not a recommendation to surgeons to remove implants in the absence of a BIA-ALCL diagnosis”.
At Absolute Cosmetic Medicine do not use Allergan branded implants. We have suspended the use of Polytech Microthane Polyurethane implants, although there have only been 2 cases of BIA-ALCL with this implant world-wide. This decision will be re-evaluated once the TGA has made its decision. We will continue to use Polyurethane Mesmo (coarse texture) and Polytxt Micro textured as neither of these implants are on any of the concern lists. These implants are seen as the safest in the world due to the low BIA-ALCL cases. We will also continue to use Polyurethane Smooth implants.
We are offering complimentary breast implant checks. At the checks, we can discuss any concerns that you have including; malposition, any pain or discomfort, capsular contracture and more. Dr Murray will also perform an ultrasound scan to check for ruptures. To book your complimentary breast implant check please call us on 93899099 or email us at reception@absolutecosmetic.com.au.
Update – The TGA has released a fact sheet titled “Recall of Allergan textured implants: what you need to know if you have implants”. See the fact sheet here.